Global Small Molecules CDMOs Mapping
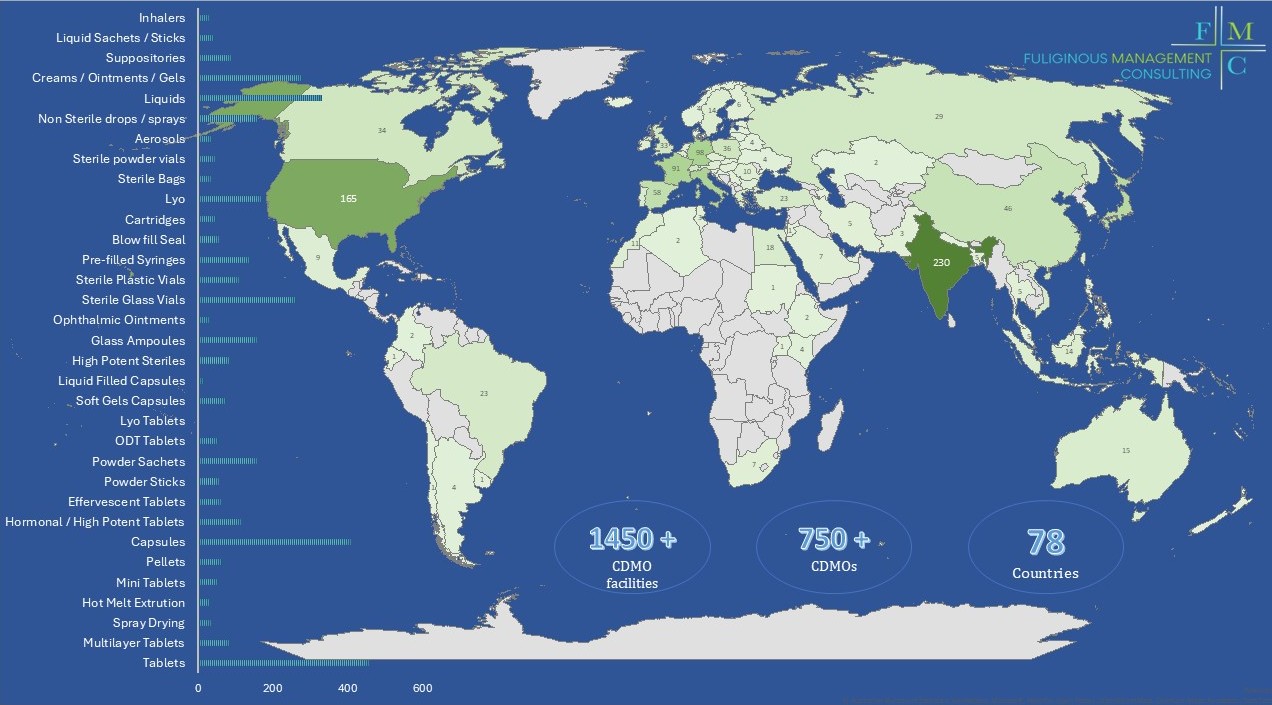
The Global Pharmaceutical CDMOs mapping contains CDMOs (or companies offering CDMO services to third parties) that focus on the production of pharmaceutical finished products and have the capability to produce commercial scale batches.
Overall it includes more than 750 CMOs/CDMOs and 1450 manufacturing sites globally.
The overall number of countries with manufacturing sites is 78.
It also includes:
- The number of manufacturing sites of each CDMO (excluding sites that do not manufacture finished dosage forms).
- The location of the manufacturing sites.
- 35 + different manufacturing technologies.
- Capabilities at Manufacturing Site Level.
- If a Manufacturing Site is FDA and EU GMP approved.
- If a CDMO offers Development services.
- If a CDMO offers API Contract Manufacturing services.
- If a CDMO has a Packaging Only Site
- If a CDMO has the capability to handle Beta lactams.
- It also contains information about the capability of the CDMOs to produce food supplements, cosmetics and VET products.
Biologics CDMOs Mapping

Apart from the fact that there are many different types of biologic products, at the moment there are not too many CDMOs able to manufacture drug substance and then produce a product, let alone that there are some CDMOs that don’t have commercial scale capabilities and some that do not have even filling capabilities (and if they have filling capabilities, not all of them can fill every type of primary packaging).
So, what we wanted to clarify for us and for our customers, is which CDMO is able to do what and up to what step. In this attempt we checked the profiles of more than 110 Bio CDMOs and we checked 11 different types of bio products as well as the main primary packaging (filling) options. Moreover, we mapped which CDMOs offer cell line development, which can offer commercial batches and which are FDA approved.
